Radhe Radhe, B.Pharm students! In this post, you will find the notes for your 8th semester Quality Control and Standardization of Herbals subject . Here you will find notes, books, previous year questions (PYQs), and important question-answers related to your pharmacy course Syllabus. You will also find GPAT updates here and exam tips. Here You will find pharmacy-related vacancies, so stay calm and connected with Pulse by Anubhav. Join Our Telegram Channel As Well As Bookmark Our Site For The Latest Update.

As you may have realized, pursuing a B.Pharm degree is not easy. Many people initially choose it as just one option, but later they discover how much studying is involved. Your B.Pharm course is divided into 8 semesters. In which the eight semester, you have to study total number of 4 subjects . In this subject you have to select the subject that you have interest and you want to follow the same in your higher study. You won’t have to study any additional subjects in this semester.
The pharmacy field is a very important medical field. Here, pharmacists not only dispense medication to patients but also educate them about the medicines. In the pharmaceutical industry, these same pharmacists manufacture the medicines and conduct research on them.
The link to download notes for all units is given below. If you want to download the PDF without reading the post, you can download the PDF by clicking on the link given below. You can also join our Telegram channel for future updates.
The Subjects in the 8th semester of B.Pharm are:
Core Subjects
- Biostatistics and Research Methodology
- Social and Preventive Pharmacy
Elective Subjects
- Pharma Marketing Management
- Pharmaceutical Regulatory Science
- Pharmacovigilance
- Quality Control and Standardization of Herbals
- Computer Aided Drug Design
- Cell and Molecular Biology
- Cosmetic Science
- Pharmacological Screening Methods
- Advanced Instrumentation Techniques
- Dietary Supplements and Nutraceuticals
- Pharmaceutical Product Development
In Quality Control and Standardization of Herbals, you will be taught how the drug delivery takes place and how the research on the medicine dose are perform.
B.Pharm 8th Semester Quality Control and Standardization of Herbals Syllabus
Before downloading the notes, please take a look at the Quality Control and Standardization of Herbals syllabus to understand what topics are covered.
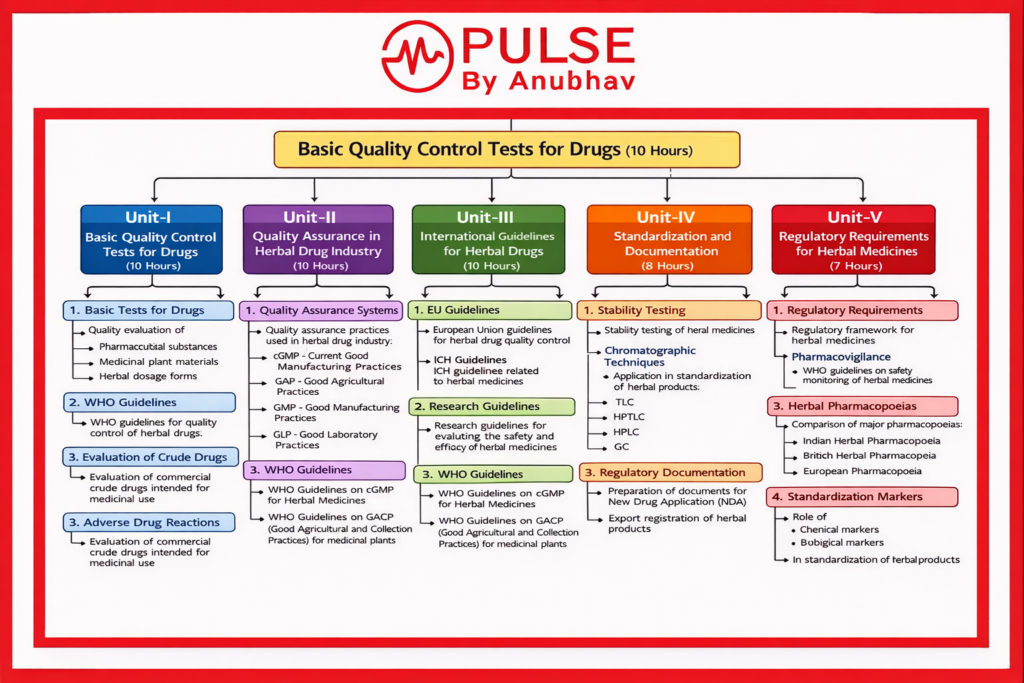
Unit I — Basic Quality Control Tests for Drugs
1. Basic Tests for Drugs
Quality evaluation of:
- Pharmaceutical substances
- Medicinal plant materials
- Herbal dosage forms
2. WHO Guidelines
- WHO guidelines for quality control of herbal drugs
3. Evaluation of Crude Drugs
- Evaluation of commercial crude drugs intended for medicinal use
Unit II — Quality Assurance in Herbal Drug Industry
1. Quality Assurance Systems
Quality assurance practices used in herbal drug industry:
- cGMP – Current Good Manufacturing Practices
- GAP – Good Agricultural Practices
- GMP – Good Manufacturing Practices
- GLP – Good Laboratory Practices
2. Traditional Systems of Medicine
- Quality assurance practices used in traditional medicines
3. WHO Guidelines
- WHO Guidelines on cGMP for Herbal Medicines
- WHO Guidelines on GACP (Good Agricultural and Collection Practices) for medicinal plants
Unit III — International Guidelines for Herbal Drugs
1. EU Guidelines
- European Union guidelines for herbal drug quality control
2. ICH Guidelines
- ICH guidelines related to herbal medicines
3. Research Guidelines
- Research guidelines for evaluating the safety and efficacy of herbal medicines
Unit IV — Standardization and Documentation
1. Stability Testing
- Stability testing of herbal medicines
2. Chromatographic Techniques
Application in standardization of herbal products:
- TLC
- HPTLC
- HPLC
- GC
3. Regulatory Documentation
- Preparation of documents for New Drug Application (NDA)
- Export registration of herbal products
4. Legal Requirements
- GMP requirements
- Drugs and Cosmetics Act provisions
Unit V — Regulatory Requirements for Herbal Medicines
1. Regulatory Requirements
- Regulatory framework for herbal medicines
2. Pharmacovigilance
- WHO guidelines on safety monitoring of herbal medicines
3. Herbal Pharmacopoeias
Comparison of major pharmacopoeias:
- Indian Herbal Pharmacopoeia
- British Herbal Pharmacopoeia
- European Pharmacopoeia
4. Standardization Markers
Role of:
- Chemical markers
- Biological markers
in standardization of herbal products
Also Read- Not Drinking Enough Water? Discover the Effects of Dehydration on Your Body
B.Pharm 8th Semester Quality Control and Standardization of Herbals Notes PDF
🌿 Download B.Pharm – Herbal Drug Quality Control Notes (PCI Syllabus | Unit-wise | Flowchart PDF)
⚡ Herbal drug quality control notes are prepared in simple exam-oriented flowchart format specially for B.Pharm students – quick revision & high scoring.
🚀 Join Pulse Pharma Notes Telegram Channel 📘 Download Complete Herbal Drug Quality Control Flowchart Notes (PDF)Also Read:- B.Pharm 7th Semester Industrial Pharmacy-2 Notes
B.Pharm 8th Semester Quality Control and Standardization of Herbals Book PDF
Books are very important; you should definitely check out the books according to your syllabus. During your pharmacy studies, books from Nirali Prakashan and PV Sindhu are very helpful. You can download the PDFs from the link below.
- Pharmaceutical Quality Control and Standardization of Herbals Nirali Prakashan Full Book PDF:- PDF
- Pharmaceutical Quality Control and Standardization of Herbals PV Sindhu Full Book PDF:- PDF
- Pharmaceutical Quality Control and Standardization of Herbals Thakur Publication Full Book PDF:- PDF
- Pharmaceutical Quality Control and Standardization of Herbals The Point Publication Full Book PDF: PDF
