You will get here Chemistry Class 12 CBSE Notes and MCQs Chapter 1: Electrochemistry . You can connect with us for every important update related to class 12.
Chemistry is a very important subject for your board exam because it will help you increase your percentage and give big support in your entrance exam like JEE and NEET. You can score high marks in this subject by studying notes and practicing MCQs. In this post, we will cover Chapter 1: Electrochemistry .
Also Read- Sophie Shine Wedding, Biography, Career & Personal Life
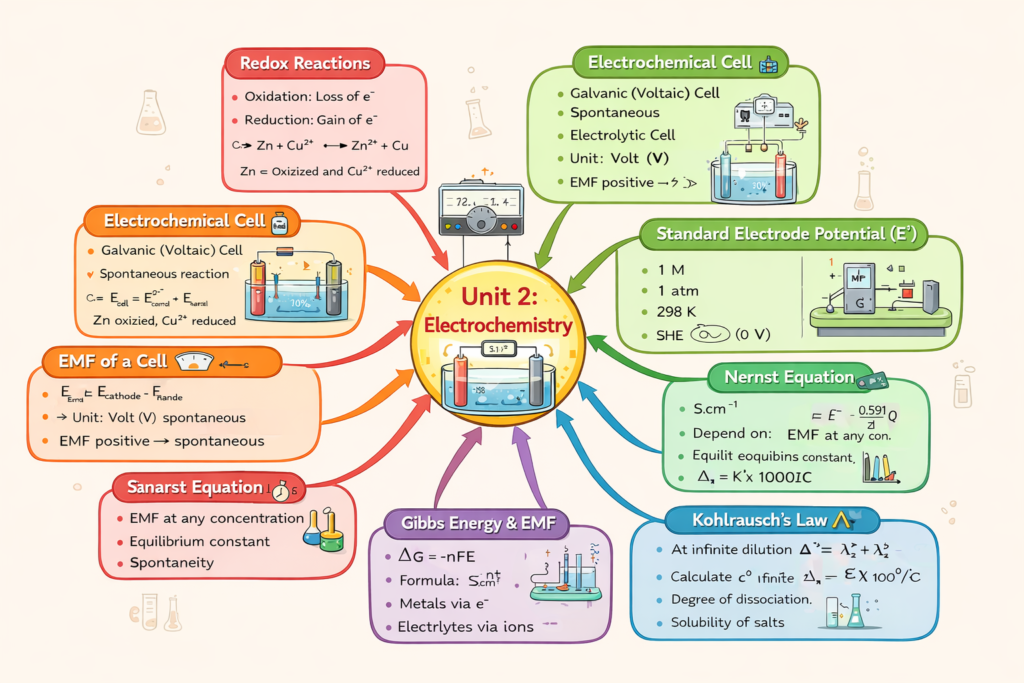
Key Content Of Chapter 2 Electrochemistry 🔹 1. Redox Reactions
CBSE Class 12 Chemistry – Unit 2
Electrochemistry (Redox, EMF, Nernst Equation, Conductivity & Electrolysis)
Complete Class 12 Chemistry Unit 2 (Electrochemistry) notes, syllabus,
NCERT book PDF aur official Telegram channel ka direct access yahan se milega.
Isme redox reactions, galvanic & electrolytic cells, EMF, standard electrode potential,
Nernst equation, Gibbs energy relation, conductance, molar conductivity,
Kohlrausch’s law, electrolysis, batteries, fuel cells aur corrosion cover hai.
Also Read- Jemimah Rodrigues: Why She’s Trending, Biography, Net Worth, Age, Lifestyle & Career 2026
Q1. Redox reactions involve:
A. Transfer of electrons
B. Transfer of neutrons
C. Transfer of protons only
D. No electron change
✅ Correct Answer: Transfer of electrons
Q2. EMF of a cell is the potential difference when:
A. No current flows
B. Maximum current flows
C. Temperature increases
D. Resistance is zero
✅ Correct Answer: No current flows
Q3. Standard electrode potential is measured relative to:
A. Standard hydrogen electrode
B. Copper electrode
C. Zinc electrode
D. Silver electrode
✅ Correct Answer: Standard hydrogen electrode
Q4. Nernst equation is used to calculate:
A. Cell potential at non-standard conditions
B. Atomic mass
C. Temperature
D. Pressure
✅ Correct Answer: Cell potential at non-standard conditions
Q5. The relation between Gibbs energy and EMF is:
A. ΔG = −nFE
B. ΔG = nFE
C. ΔG = RT
D. ΔG = PV
✅ Correct Answer: ΔG = −nFE
Q6. Conductance of electrolytic solution is due to:
A. Movement of ions
B. Movement of neutrons
C. Chemical bonding
D. Temperature only
✅ Correct Answer: Movement of ions
Q7. Molar conductivity increases with dilution because:
A. Degree of ionisation increases
B. Temperature decreases
C. Pressure increases
D. Resistance increases
✅ Correct Answer: Degree of ionisation increases
Q8. Kohlrausch’s law states that:
A. Limiting molar conductivity is sum of ionic conductivities
B. Conductivity increases with pressure
C. Resistance is zero
D. Current stops at equilibrium
✅ Correct Answer: Limiting molar conductivity is sum of ionic conductivities
Q9. In electrolysis, energy conversion is:
A. Electrical to chemical
B. Chemical to electrical
C. Heat to electrical
D. Light to chemical
✅ Correct Answer: Electrical to chemical
Q10. Rusting of iron is an example of:
A. Corrosion
B. Reduction
C. Neutralisation
D. Sublimation
✅ Correct Answer: Corrosion
Chemistry Chapter 2 Electrochemistry Quick Revision 🔹 Redox Reactions (Concept)
🔹 Electrochemical Cell
🔹 EMF of a Cell
🔹 Standard Electrode Potential (E°)
🔹 Nernst Equation
🔹 Relation Between Gibbs Energy and EMF
🔹 Conductance in Electrolytic Solutions
🔹 Specific Conductivity (κ)
🔹 Molar Conductivity (Λm)
🔹 Variation of Conductivity with Concentration
🔹 Kohlrausch’s Law
🔹 Electrolysis
🔹 Laws of Electrolysis
Also Read- CBSE Class 12 Chemistry Chapter 1: Solutions – Notes + MCQs | Quick Revision
Electrochemistry FAQs About Chapter Electrochemistry
What is Electrochemistry?
Electrochemistry chemical energy aur electrical energy ke conversion ka study hai jisme redox reactions involved hote hain.
What is a Galvanic Cell?
Galvanic cell spontaneous redox reaction se chemical energy ko electrical energy me convert karta hai.
What is an Electrolytic Cell?
Electrolytic cell me electrical energy use karke non-spontaneous chemical reaction karwaya jata hai.
What is EMF of a Cell?
EMF cell ke electrodes ke beech potential difference hota hai jo electron flow ki driving force hota hai.
What is Standard Electrode Potential?
Standard conditions (1M, 1 atm, 298K) par electrode ka potential standard electrode potential kehlata hai.
What is Nernst Equation?
Nernst equation non-standard conditions par electrode ya cell potential calculate karne ke liye use hota hai.
What is Molar Conductivity?
1 mole electrolyte ki conductance ko molar conductivity kehte hain aur dilution se increase hoti hai.
What is Kohlrausch’s Law?
Infinite dilution par electrolyte ki conductivity individual ions ki conductivity ke sum ke equal hoti hai.
What is Electrolysis?
Electric current se chemical reaction karwana electrolysis kehlata hai.
What is Corrosion?
Metal ka environment ke saath reaction karke degrade hona corrosion kehlata hai (jaise iron rusting).
Unit 8: Aldehydes, Ketones and Carboxylic Acids
View Post Unit 7: Alcohols, Phenols and Ethers
View Post 

1 thought on “CBSE Class 12 Chemistry Chapter 2: Electrochemistry – Notes + MCQs | Quick Revision”