You will get here Chemistry Class 12 CBSE Notes and MCQs Chapter 9: Amines. You can connect with us for every important update related to class 12.

Chemistry is a very important subject for your board exam because it will help you increase your percentage and give big support in your entrance exam like JEE and NEET. You can score high marks in this subject by studying notes and practicing MCQs. In this post, we will cover Chapter 9: Amines .
Also Read- Sophie Shine Wedding, Biography, Career & Personal Life
Key Content Of Chapter 9 Amines
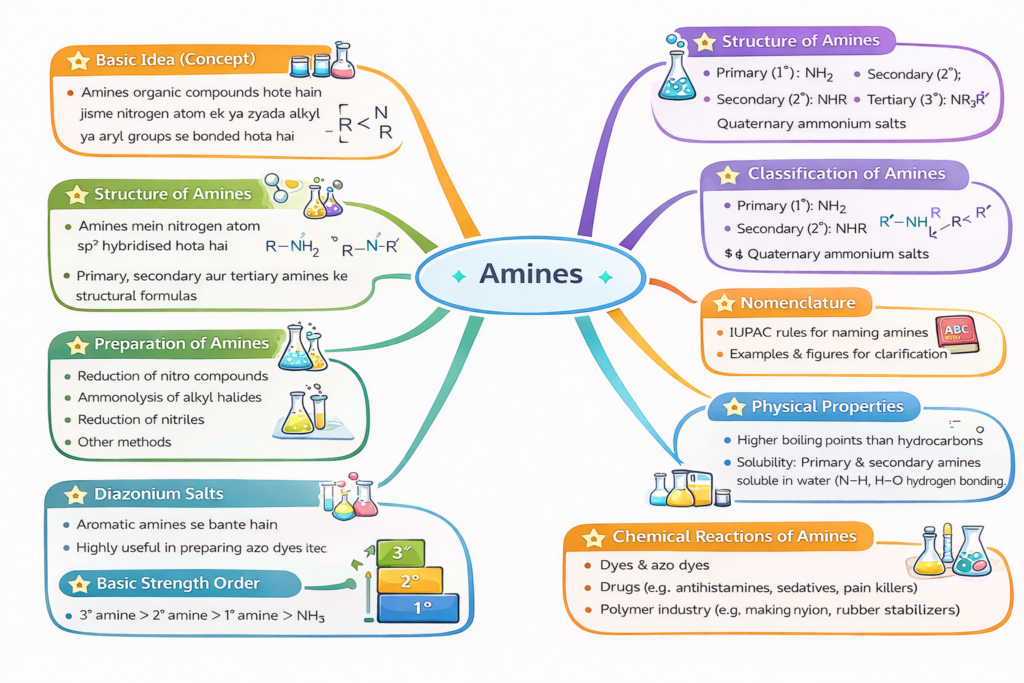
🔹 Basic Idea (Concept)
🔹 Structure of Amines
🔹 Classification of Amines
🔹 Nomenclature
🔹 Preparation of Amines
🔹 Physical Properties
🔹 Chemical Reactions of Amines
🔹 Basic Strength Order
⭐ Diazonium Salts
⭐ Commercial Uses of Amines
Class 12 Chemistry Chapter 9: Amines Notes
Also Read- Jemimah Rodrigues: Why She’s Trending, Biography, Net Worth, Age, Lifestyle & Career 2026
Chemistry Chapter 9 Amines MCQs
Chemistry Chapter 9 Amines Quick Revision
🔹 Basic Idea (Real Life + Concept)
Amines ammonia (NH₃) ke derivatives hote hain jisme hydrogen atoms alkyl ya aryl groups se replace ho jaate hain. Nitrogen par lone pair hone ki wajah se amines basic nature show karte hain aur medicines, dyes aur fibres me use hote hain.
🔹 Structure of Amines
Amines me nitrogen atom sp³ hybridised hota hai aur molecule pyramidal shape show karta hai. Lone pair repulsion ki wajah se bond angle 109.5° se thoda kam (~108°) hota hai.
🔹 Classification of Amines
Amines ko hydrogen replacement ke basis par classify kiya jata hai:
- Primary amine (1°) → R–NH₂
- Secondary amine (2°) → R₂NH
- Tertiary amine (3°) → R₃N
Same groups → simple amines
Different groups → mixed amines.
🔹 Nomenclature
Amines ko common aur IUPAC system se name kiya jata hai.
- Common name: Alkyl + amine (methylamine)
- IUPAC name: Alkane → “e” replace by “amine” (methanamine)
- Secondary aur tertiary me N- prefix use hota hai.
- Aromatic amine example → Aniline (Benzenamine).
🔹 Preparation of Amines
Amines banane ke main methods:
- Nitro compounds ka reduction → amines
- Alkyl halides ka ammonolysis
- Nitriles ka reduction
- Amides ka reduction
- Gabriel phthalimide synthesis (primary amine)
- Hoffmann bromamide reaction (one carbon less amine)
🔹 Physical Properties
- Lower amines gases hote hain (fishy smell).
- Higher amines liquids ya solids hote hain.
- Lower amines water soluble hote hain (hydrogen bonding).
- Solubility chain length badhne par decrease hoti hai.
- Boiling point order: Primary > Secondary > Tertiary.
🔹 Chemical Reactions of Amines
✔ Basic Nature
Amines acids ke saath salt banate hain.
✔ Alkylation
Amines alkyl halides se react karke higher amines banate hain.
✔ Acylation
Amines acyl chloride se react karke amides banate hain.
✔ Carbylamine Reaction
Primary amines chloroform + KOH ke saath foul smell isocyanide banate hain (test for 1° amine).
✔ Reaction with Nitrous Acid
Primary, secondary aur tertiary amines different products dete hain.
✔ Electrophilic Substitution
Aromatic amines me –NH₂ group ortho-para directing hota hai.
🔹 Basic Strength Order
Amines ki basic strength electron density aur structure par depend karti hai.
- Aliphatic amines > Ammonia > Aromatic amines
- Aqueous solution me order generally:
Secondary > Primary > Tertiary > NH₃ - Aromatic amines resonance ki wajah se weak base hote hain.
⭐ Diazonium Salts
Aromatic primary amines nitrous acid se react karke diazonium salts banate hain (273–278 K).
Ye aromatic compounds me different groups introduce karne aur azo dyes banane me use hote hain.
⭐ Commercial Uses of Amines
- Medicines aur pharmaceuticals
- Dyes aur polymers
- Synthetic fibres
- Agrochemicals
- Insect attractants
- Chemical synthesis intermediates

1 thought on “CBSE Class 12 Chemistry Chapter 9: Amines – Notes + MCQs | Quick Revision”